
Simultaneous deflections by applied electric and magnetic fields permitted Thomson to calculate the mass-to-charge ratio of the particles composing the cathode ray. (c) In the cathode ray, the beam (shown in yellow) comes from the cathode and is accelerated past the anode toward a fluorescent scale at the end of the tube. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. Thomson produced a visible beam in a cathode ray tube. The results of these measurements indicated that these particles were much lighter than atoms (Figure 1).įigure 1. In similar experiments, the ray was simultaneously deflected by an applied magnetic field, and measurements of the extent of deflection and the magnetic field strength allowed Thomson to calculate the charge-to-mass ratio of the cathode ray particles. This beam was deflected toward the positive charge and away from the negative charge, and was produced in the same way with identical properties when different metals were used for the electrodes. When high voltage was applied across the electrodes, a visible beam called a cathode ray appeared between them. This apparatus consisted of a sealed glass tube from which almost all the air had been removed the tube contained two metal electrodes.

If matter were composed of atoms, what were atoms composed of? Were they the smallest particles, or was there something smaller? In the late 1800s, a number of scientists interested in questions like these investigated the electrical discharges that could be produced in low-pressure gases, with the most significant discovery made by English physicist J. Atomic Theory after the Nineteenth Century While the historical persons and dates behind these experiments can be quite interesting, it is most important to understand the concepts resulting from their work. Here, we will discuss some of those key developments, with an emphasis on application of the scientific method, as well as understanding how the experimental evidence was analyzed. Much of this came from the results of several seminal experiments that revealed the details of the internal structure of atoms. In the two centuries since Dalton developed his ideas, scientists have made significant progress in furthering our understanding of atomic theory.

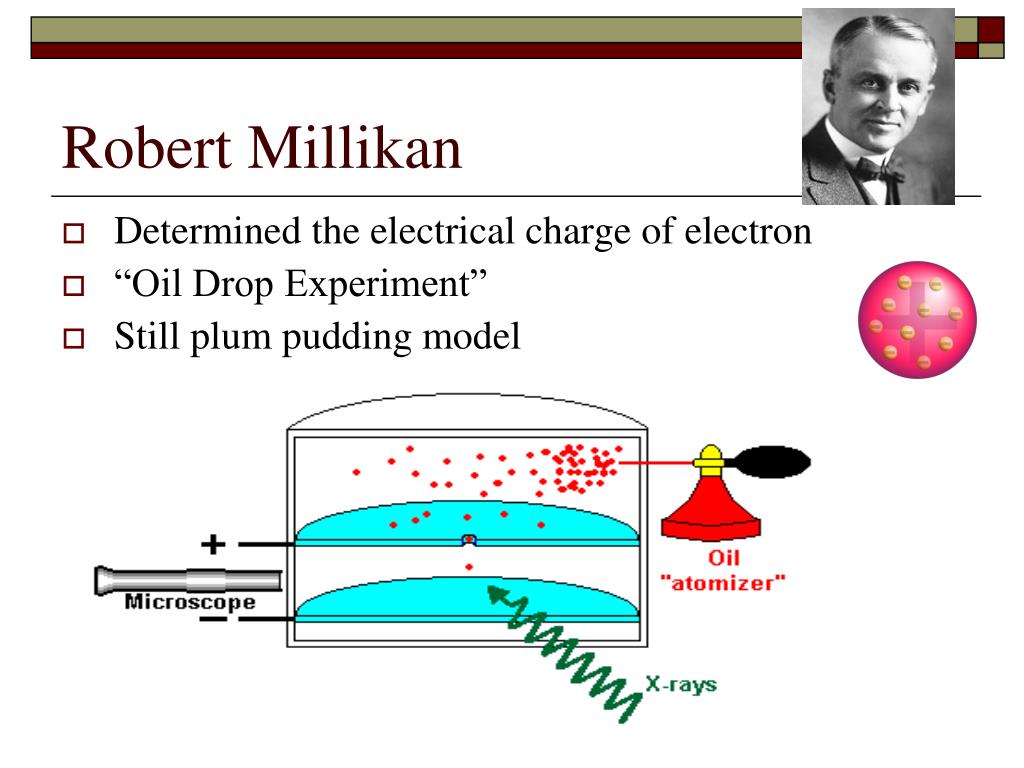
During 1920-1923, Millikan occupied himself with work concerning the hot-spark spectroscopy of the elements (which explored the region of the spectrum between the ultraviolet and X-radiation), thereby extending the ultraviolet spectrum downwards far beyond the then known limit. In addition, his studies of the Brownian movements in gases put an end to all opposition to the atomic and kinetic theories of matter. Next, he verified experimentally Einstein's all-important photoelectric equation, and made the first direct photoelectric determination of Planck's constant h (1912-1915). His earliest major success was the accurate determination of the charge carried by an electron, using his "falling-drop method" he also proved that this quantity was a constant for all electrons (1910), thus demonstrating the atomic structure of electricity. Millikan made numerous discoveries in the fields of electricity, optics, and molecular physics. During his early years at Chicago, he spent much time preparing textbooks and simplifying the teaching of physics. Millikan was an eminent teacher, and passing through the customary grades he became professor at that university in 1910, a post which he retained until 1921. Michelson, to become assistant at the newly established Ryerson Laboratory at the University of Chicago. Millikan spent a postgraduate year (from 1895-1896) in Germany, at the Universities of Berlin and Gottingen. In 1893, after obtaining his mastership in physics, he was appointed Fellow in Physics at Columbia University. It was during this period that he developed his interest in the subject in which he was later to excel. During his undergraduate course at Oberlin College, his favorite subjects were Greek and mathematics but after his graduation in 1891 he took, for two years, a teaching post in elementary physics. Robert Andrews Millikan was born in Morrison, Illinois, in 1868.

